 4 The students are considered to be colearners with the teacher, both learning from the interview/feedback. 1, which represents a layer of ions in NaCl. Most of the comments discussed in this article relate to Fig. The main research technique is a sequence of in-depth interviews, focused on figures showing atoms, molecules, lattices etc. I have a particular interest in studying students' understanding of chemical bonding and its development during an A-level course. 
Despite a large literature covering many aspects of school science there has been little attention focused on the topic of bonding. Researchers have used a variety of terms to describe such alternative ideas – such as misconceptions, alternative conceptions, children's science, alternative frameworks, intuitive theories, informal ideas, theories in action, and mini theories. The learner's own ideas may be fragmentary and only weakly held to, or they may comprise an extensive and coherent frame- work of concepts that has much explanatory value for the learner. 2 The viewpoint underlying such research is that learners often construct their own meanings and understandings, based as much on what they experience outside the classroom as within – but much of this 'knowledge' is actually at variance with accepted science, and can impede orthodox understanding. For example, the Children's Learning in Science Project (CLiSP) has produced materials about understanding particle theory 1 and basic chemistry. There has been a great deal of research into children's ideas and understanding in a number of scientific topic areas. If these common misconceptions are more than just coincidence, and are found to be wide- spread, then there could be implications for the teaching of chemistry in schools, colleges and sixth forms. Moreover, there seemed to be similarities between the wrong ideas revealed – even though the students concerned came to Havering College from different schools. One aspect of particular interest was that, although these students were able to discuss ionic bonding, many of their comments implied that their mental models were incorrect. 
As might be expected, there were variations in the breadth of knowledge and depth of understanding. 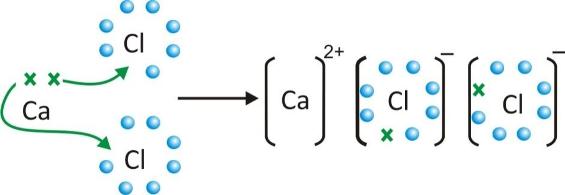
The 10 'volunteers' were inter- viewed during the first few weeks of term to find out how much they knew and under- stood about chemical bonding at the start of the course. Could there be implications for the way in which bonding is taught at Key Stage 4 and A-level?Īt the beginning of their A-level chemistry course, I invited some of my students to take part in a study of how their understanding of chemical bonding developed during the course. 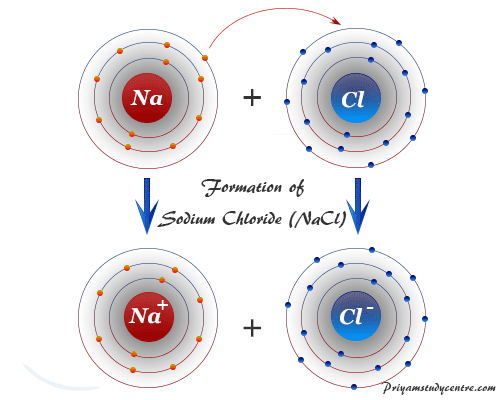
The text of the article follows: Misunderstanding the ionic bond Students beginning A-level chemistry seem to have misconceptions about the nature of ionic bonding. (For those with access, the pdf can be downloaded from. Content of older issues of Education in Chemistry has been scanned and included in the Royal Society of Chemistry's historical on-line archive, available to members, but not freely available to the general public. (1994) Misunderstanding the ionic bond, Education in Chemistry, 31 (4), pp.100-103.Īlthough Education in Chemistry has a web-site which includes recent articles form the magazine, this does not go back as far as 1994. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
